Articles
- Page Path
- HOME > Acute Crit Care > Volume 32(1); 2017 > Article
-
Review
Surgery Management of Critical Burn Injuries: Recent Developments -
David J. Dries1
 , John J. Marini2
, John J. Marini2 -
Korean Journal of Critical Care Medicine 2017;32(1):9-21.
DOI: https://doi.org/10.4266/kjccm.2016.00969
Published online: February 17, 2017
1Department of Surgery and Anesthesiology, University of Minnesota, Minneapolis, MN, USA
2Department of Medicine, University of Minnesota, Minneapolis, MN, USA
- Correspondence to: David J. Dries, Department of Surgery, Regions Hospital, 640 Jackson Street, St. Paul, MN 55101, USA Tel: +1-651-254-1633, Fax: +1-651-254-1480 E-mail: david.j.dries@healthpartners.com
• Received: December 2, 2016 • Accepted: December 25, 2016
Copyright © 2017 The Korean Society of Critical Care Medicine
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
-
Background
- Burn injury and its subsequent multisystem effects are commonly encountered by acute care practitioners. Resuscitation is the major component of initial burn care and must be managed to restore and preserve vital organ function. Later complications of burn injury are dominated by infection. Burn centers are often called to manage problems related to thermal injury, including lightning and electrical injuries.
-
Methods
- A selected review is provided of key management concepts as well as of recent reports published by the American Burn Association.
-
Results
- The burn-injured patient is easily and frequently over resuscitated, with ensuing complications that include delayed wound healing and respiratory compromise. A feedback protocol designed to limit the occurrence of excessive resuscitation has been proposed, but no new “gold standard” for resuscitation has replaced the venerated Parkland formula. While new medical therapies have been proposed for patients sustaining inhalation injury, a paradigm-shifting standard of medical therapy has not emerged. Renal failure as a specific contributor to adverse outcome in burns has been reinforced by recent data. Of special problems addressed in burn centers, electrical injuries pose multisystem physiologic challenges and do not fit typical scoring systems.
-
Conclusion
- Recent reports emphasize the dangers of over resuscitation in the setting of burn injury. No new medical therapy for inhalation injury has been generally adopted, but new standards for description of burn-related infections have been presented. The value of the burn center in care of the problems of electrical exposure, both manmade and natural, is demonstrated in recent reports.
Introduction
Resuscitation
Sepsis and Infection in Burn-Injured Patients
Electrical and Lightning Injury
Pulmonary Issues
Outcomes
Conclusions
Acknowledgments
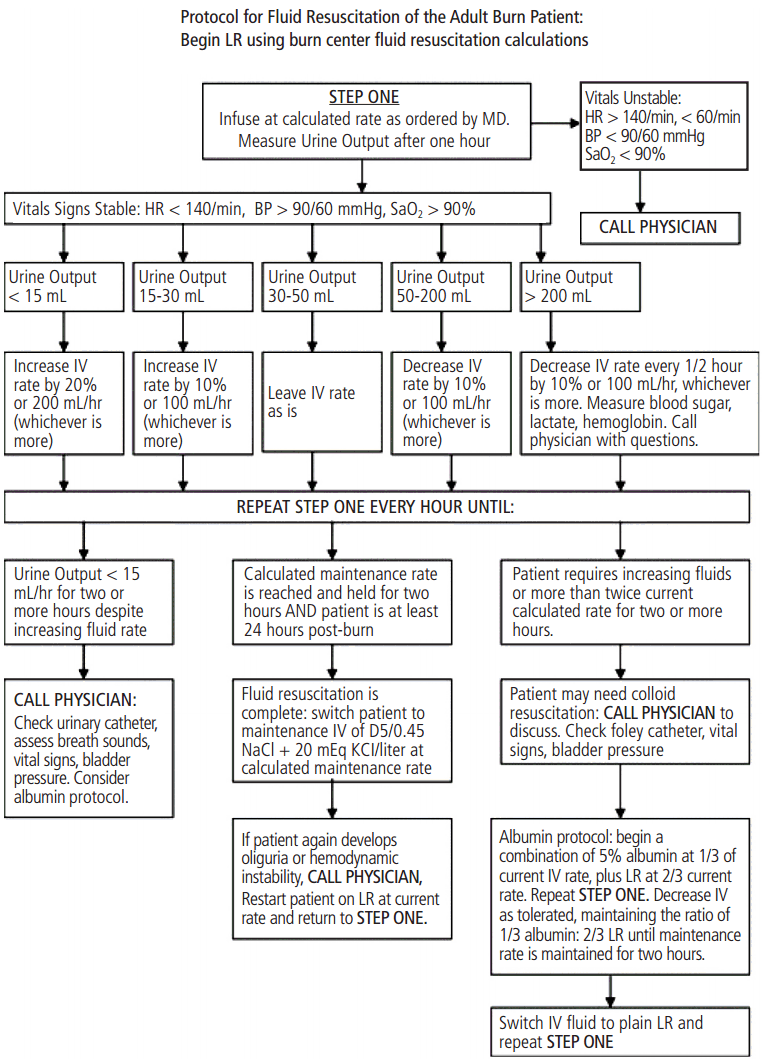
Figure 1.Protocol for fluid resuscitation of adult burn patients. In response to requests from nurses, this protocol was developed to permit nursing staff to manage fluid resuscitation of acute burn patients. Initial fluid rates are calculated by the Parkland formula. Nurses begin hourly infusion, measure urine output, and adjust fluids according to patient response. Development of unstable vital signs, inadequate response to fluids, or persistently high fluid requirements prompt a call to the physician. A pathway to begin colloid replacement exists for patients who display increasing fluid requirements or develop symptoms of torso compartment syndrome. Adopted from references [3]. with permission. LR: lactated Ringer’s solution; D5: 5% dextrose.

- 1. Latenser BA. Critical care of the burn patient: the first 48 hours. Crit Care Med 2009;37:2819-26.ArticlePubMed
- 2. Baxter CR. Fluid volume and electrolyte changes of the early postburn period. Clin Plast Surg 1974;1:693-703.ArticlePubMed
- 3. Saffle JR. The phenomenon of “fluid creep” in acute burn resuscitation. J Burn Care Res 2007;28:382-95.ArticlePubMed
- 4. Pham TN, Cancio LC, Gibran NS, American Burn Association. American Burn Association practice guidelines burn shock resuscitation. J Burn Care Res 2008;29:257-66.ArticlePubMed
- 5. Blumetti J, Hunt JL, Arnoldo BD, Parks JK, Purdue GF. The Parkland formula under fire: is the criticism justified? J Burn Care Res 2008;29:180-6.ArticlePubMed
- 6. Greenhalgh DG, Saffle JR, Holmes JH 4th, Gamelli RL, Palmieri TL, Horton JW, et al. American Burn Association consensus conference to define sepsis and infection in burns. J Burn Care Res 2007;28:776-90.ArticlePubMed
- 7. Uchino S, Kellum JA, Bellomo R, Doig GS, Morimatsu H, Morgera S, et al. Acute renal failure in critically ill patients: a multinational, multicenter study. JAMA 2005;294:813-8.ArticlePubMed
- 8. Koumbourlis AC. Electrical injuries. Crit Care Med 2002;30(11 Suppl):S424-30.ArticlePubMed
- 9. Ivy ME, Atweh NA, Palmer J, Possenti PP, Pineau M, D’Aiuto M. Intra-abdominal hypertension and abdominal compartment syndrome in burn patients. J Trauma 2000;49:387-91.ArticlePubMed
- 10. Bone RC, Balk RA, Cerra FB, Dellinger RP, Fein AM, Knaus WA, et al. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest 1992;101:1644-55.ArticlePubMed
- 11. Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, et al. Surviving sepsis guidelines: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med 2013;41:580-637.ArticlePubMed
- 12. Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 2016;315:801-10.ArticlePubMedPMC
- 13. Hubmayr RD, Burchardi H, Elliot M, Fessler H, Georgopoulos D, Jubran A, et al. Statement of the 4th international consensus conference in critical care on ICU-acquired pneumonia--Chicago, Illinois, May 2002. Intensive Care Med 2002;28:1521-36.ArticlePubMed
- 14. Rello J, Paiva JA, Baraibar J, Barcenilla F, Bodi M, Castander D, et al. International conference for the development of consensus on the diagnosis and treatment of ventilator-associated pneumonia. Chest 2001;120:955-70.ArticlePubMed
- 15. Mosier MJ, Pham TN. American Burn Association practice guidelines for prevention, diagnosis, and treatment of ventilator-associated pneumonia (VAP) in burn patients. J Burn Care Res 2009;30:910-28.ArticlePubMed
- 16. Sen S, Johnston C, Greenhalgh D, Palmieri T. Ventilator-associated pneumonia prevention bundle significantly reduces the risk of ventilator-associated pneumonia in critically ill burn patients. J Burn Care Res 2016;37:166-71.ArticlePubMed
- 17. Chung KK, Rhie RY, Lundy JB, Cartotto R, Henderson E, Pressman MA, et al. A survey of mechanical ventilator practices across burn centes in North America. J Burn Care Res 2016;37:e131-9.ArticlePubMedPMC
- 18. Peck MD, Weber J, McManus A, Sheridan R, Heimbach D. Surveillance of burn wound infections: a proposal for definitions. J Burn Care Rehabil 1998;19:386-9.ArticlePubMed
- 19. Arnoldo BD, Purdue GF, Kowalske K, Helm PA, Burris A, Hunt JL. Electrical injuries: a 20-year review. J Burn Care Rehabil 2004;25:479-84.ArticlePubMed
- 20. Lichtenberg R, Dries D, Ward K, Marshall W, Scanlon P. Cardiovascular effects of lightning strikes. J Am Coll Cardiol 1993;21:531-6.ArticlePubMed
- 21. Arnoldo B, Klein M, Gibran NS. Practice guidelines for the management of electrical injuries. J Burn Care Res 2006;27:439-47.ArticlePubMed
- 22. Singerman J, Gomez M, Fish JS. Long-term sequelae of low-voltage electrical injury. J Burn Care Res 2008;29:773-7.ArticlePubMed
- 23. ARDS Definition Task Force; Ranieri VM, Rubenfeld GD, Thompson BT, Ferguson ND, Caldwell E, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA 2012;307:2526-33.PubMed
- 24. Dries DJ, Endorf FW. Inhalation injury: epidemiology, pathology, treatment strategies. Scand J Trauma Resusc Emerg Med 2013;21:31. ArticlePubMedPMC
- 25. Jeng JC. Patrimonie de Docteur Baux--Baux Scores >> 100 gleaned from 170,791 admissions: a glimmer from the National Burn repository. J Burn Care Res 2007;28:380-1.ArticlePubMed
- 26. American Burn Association. 2016 National Burn Repository. Report of data from 2006-2015 [Internet]. Chicago, American Burn Association. c2017;[cited 2017 Jan 12]. Available from: www.ameriburn.org/NBR.php.
- 27. Ryan CM, Schoenfeld DA, Thorpe WP, Sheridan RL, Cassem EH, Tompkins RG. Objective estimates of the probability of death from burn injuries. N Engl J Med 1998;338:362-6.ArticlePubMed
- 28. Paratz JD, Lipman J, Boots RJ, Muller MJ, Paterson DL. A new marker for sepsis post burn injury?*. Crit Care Med 2014;42:2029-36.ArticlePubMed
- 29. Yang HT, Yim H, Cho YS, Kym D, Hur J, Kim JH, et al. Assessment of biochemical markers in the early post-burn period for predicting acute kidney injury and mortality inpatients with major burn injury: comparison of serum creatinine, serum cystatiC, plasma and urine neutrophil gelatinase-associated lipocalin. Crit Care 2014;18:R151. ArticlePubMedPMC
- 30. Palmieri TL, London JA, O’Mara MS, Greenhalgh DG. Analysis of admissions and outcomes in verified and nonverified burn centers. J Burn Care Res 2008;29:208-12.ArticlePubMed
- 31. Ehrlich PF, Rockwell S, Kincaid S, Mucha P Jr. American College of Surgeons, Committee on Trauma Verification Review: does it really make a difference? J Trauma 2002;53:811-6.ArticlePubMed
- 32. Sheridan RL. Burn care: results of technical and organizational progress. JAMA 2003;290:719-22.ArticlePubMed
References
Figure & Data
References
Citations
Citations to this article as recorded by 

- Prevalence and Trend of Depression in Burn Survivors: A Single Center Cohort Study
V S Ranganath, Smitha Segu, B S Girish, Joel M Johns, C S Meghana
Journal of Burn Care & Research.2024;[Epub] CrossRef - SUBMICROSCOPIC CHANGES IN THE HEMOCAPILLARIES OF THE CEREBRAL HEMISCLE CAUSES CAUSED BY THERMAL BURN
H. V. Lukyantseva, V. A. Pastukhova, O. I. Kovalchuk
Bulletin of Problems Biology and Medicine.2021; 3(1): 268. CrossRef - Kefir Accelerates Burn Wound Healing Through Inducing Fibroblast Cell Migration In Vitro and Modulating the Expression of IL-1ß, TGF-ß1, and bFGF Genes In Vivo
Ahmad Oryan, Esmat Alemzadeh, Mohammad Hadi Eskandari
Probiotics and Antimicrobial Proteins.2019; 11(3): 874. CrossRef - Viable placental allograft as a biological dressing in the clinical management of full-thickness thermal occupational burns
Eric L. Johnson, Elisabet K. Tassis, Georgina M. Michael, Susan G. Whittinghill
Medicine.2017; 96(49): e9045. CrossRef

 KSCCM
KSCCM
 PubReader
PubReader ePub Link
ePub Link Cite
Cite